To begin investigating a building science problem I strongly suggest you go to a search engine / images, and see just what the problem is in the eyes of the public. Try it yourself: ice damming. You will find that, unlike a decade or two ago, there are only few photos of ice dams (and many of them have been kicking around for decades). But the web is lousy with diagrams of ice damming on roof edges. Half of those diagrams are good, half are laughably bad. If the photos of actual ice dams on roofs are few, and old, then maybe ice damming is a problem that building science has already solved, deserving of a victory lap. Maybe it’s a problem that global warming has solved, or will solve, in a few years. But just in case ice damming still occurs, let’s take a look.

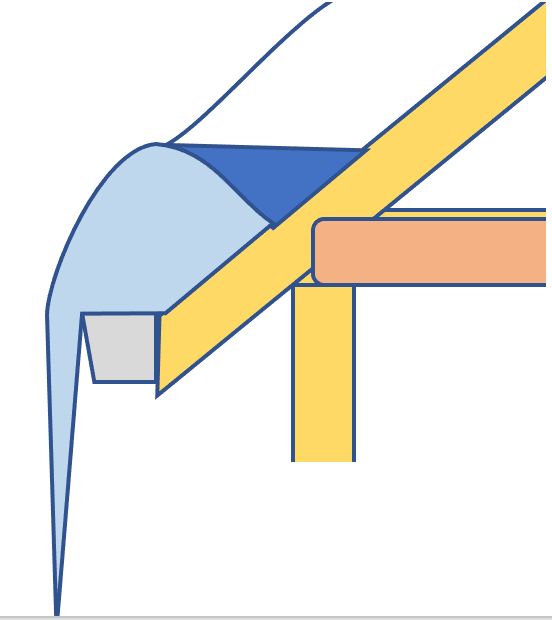
As for those images. The correct ones are based on research by Canada Mortgage and Housing Corporation (CMHC), in particular by Don Fugler. CMHC showed that in houses where ice buildup on the eaves leads to water leakage in the house, ice is found not only down at the bottom. An ice field also extends several feet (meters) up the slope of the roof, and when snow melts above the ice field it gets trapped between the deck and the ice, and builds up a head of water in that snow-ice-water-shingle-deck sandwich strong enough to force water through decking joints. The incorrect ones show a puddle of water above the wall plate, where geometry and gravity leave you wondering just how water gets from here to there.
Here are the main takeaways from thinking this problem through, to be discussed below:
- There must be a strong heat source to melt this much snow into water.
- Conductive heat loss through insulation never provides ice-dam-producing levels of heat. It needs a chimney or a big hole leaking house air.
- Ventilation essentially plays no role in ice dam formation or relief.
- Valleys are most at risk because they funnel water from a large expanse of roof area.
One group that studied ice damming was the group around Wayne Tobiasson at the Cold Regions Research and Engineering Laboratory (CRREL) of the US Army Corps of Engineers. They noted that ice dams seem to occur when the outdoor temperature is around 20 degrees F (-6.7 degrees C), a mighty helpful finding. A temperature of 20F is warm enough to get melting in many cases, and cold enough to get refreezing at the eaves. They also reached conclusions about ventilation that I challenge. See for yourself.
In building science we run the numbers. Inch-Pound units are used here, primarily because the issue of ice dams and venting to prevent them is a discussion more suited to barrooms than laboratories. Barrooms in the USA are all I-P.
We begin with constants. See the table below. A Btu is about the heat of a kitchen match. Shortened names are given to each of these constants, to appear in calculations at the end. One constant, that is not so constant, is the thermal resistivity of snow. Thermal resistivity is the thermal resistance per inch of thickness. A resistivity of one R per inch is a mid value.

Input values are shown in red.

The next table shows calculations. The important assumption in this calculation is: all of the heat comes into the attic by conduction across the thermal insulation. Each calculation is a step toward the value at the bottom, shown in blue. That is the rate at which water is formed from snow. With the inputs selected here, the amount of water formed which can turn to ice, is 2 cubic feet per day.
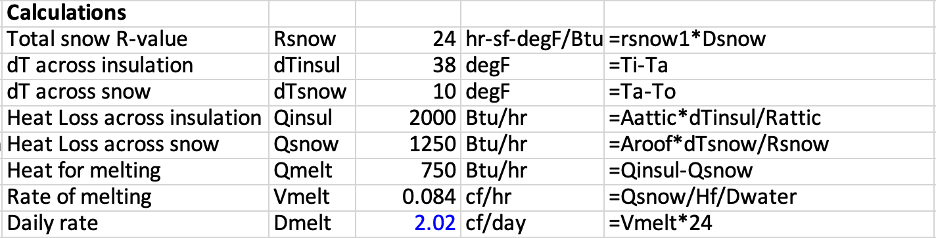
If the ice event you see shows more than 2 cubic feet per day, and that’s for the entire roof, then the conduction assumption does not hold. Which is my main point. Ice dam events that make the news, with giant icicles hanging down from the roof, are always associated with some strong heat source melting the snow, usually a chimney or hot flue, but maybe a large air leak across the ceiling large enough to warm the attic, or uninsulated leaky ductwork. Such heat losses into the attic are also money losses. Solution: get rid of the heat source.

Now to ventilation. Suppose we want to move outdoor air through the attic. What volume rate of airflow must we provide through the roof vent section area in order to chill the attic down to freezing temperature to bring the melting rate to zero? The calculations show 65 cfm. That is a lot of air for a passive vent, and it might get loud.
Note that this rush of air would have to go in and out of the attic, and at 24 inches of snowfall, the roof vents are probably snowed shut, leaving only soffit and gable vents. It might keep you awake at night. If the heat source is greater than just conductive loss, well, the volume of airflow needed depends on the strength of that heat source. Perhaps it’s calculable, it would be enormous, but who cares? Stop the heat source, period.
We are improving our attics. We insulate them heavily. We scout out air leaks across the ceiling and foam them shut. We take great care with duct placement. We try to keep heating and cooling equipment out of the attic. We have fewer heat losses from flues, thanks to greater furnace efficiency.
We tend to vent our attics, as a legacy practice. In this post I am not arguing to vent, or not to vent, or to half vent, or kinda vent. I’ll argue later when we know more about the subject. For now—please—do not use preventing ice damming as an excuse for needing to vent. If you do, then at least take the time to run the numbers using your own input values. Poe said “Believe nothing you hear, and half that you see.” As for calculations, I put them up there with observations, maybe half.

