In 1994 Dean Flessner, a vice-president of State Farm Insurance, walked into our office and asked if we could solve the problem of pipe bursting due to freezing. We learned from him that the insurance industry had four categories of claims of damage to homes—downright Aristotelean—those due to earth, air, fire and water. The insurance industry had an estimate at that time that $4B had been paid out in claims in the previous 10 years for damage from pipe bursting due to freezing.
Sure. No problem.
I decided we needed to write a beautifully illustrated guide for designers and builders on just how to keep water pipes within the insulated space. Mr. Flessner had mentioned that there were many more claims from the southern US than up north, which suggests only that northern plumbers are continually reminded of the importance of putting the pipes within the insulated space. Southern plumbers don’t have their institutional memory recharged.
Jeff Gordon, who had the lead on the project, went out and bought a freezer. Jeff knew from his weatherization contacts an Alaskan John Zarling, a researcher with considerable insight into building problems. John humbly suggested we measure water pressure.
Jeff set off to measure temperature and pressure in pipes in the freezer. This was not very easy to do when the pressure begins to build. It’s possible to create a tight gasket for the thermocouple wire as it passes into the pipe. Not easy but possible. One morning he went to the datalogger where the thermocouple wires were attached and found the wiring panel covered with water. The pressure had pushed water in between the thermocouple metal and the thin insulating jacket, all the way to the logger.
Once the measurement difficulties were overcome, he ran the test, and came to me saying that everything I know about burst water pipes due to freezing is wrong. Your thinking is probably the same as mine was. I thought that since ice has a greater volume for the mass (lower density) than water, the ice pushes against the wall of the pipe, to rupture. Wrong.
Here is the sequence of ice formation that Jeff found. First, the freezer temperature declined, and even went to around -4 degrees C. Then, in a flash, dendritic ice forms in the water, and the heat of fusion brings the temperature of now two-phase water-ice mix to exactly 0 degrees C, as it should. Then ice begins to grow from the wall of the pipe inward. This applies no stress whatsoever to the pipe wall, in fact it thickens and reinforces the pipe wall, now made of ice plus metal. This continues until the annular ice growth reaches the center of the pipe and blocks any water flow. At this point, the temperature of the now single ice phase may start going below 0C. And as ice grows, it grows along the length of the pipe.
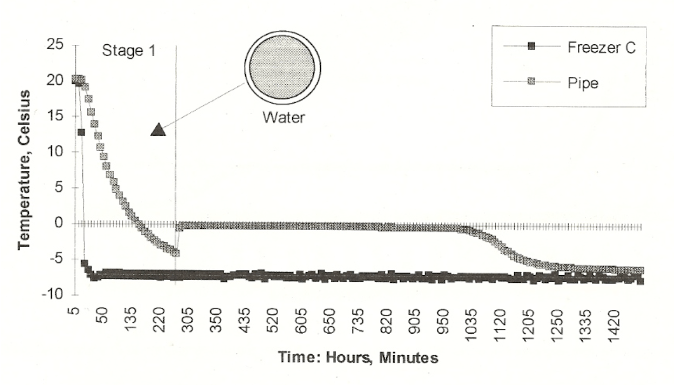
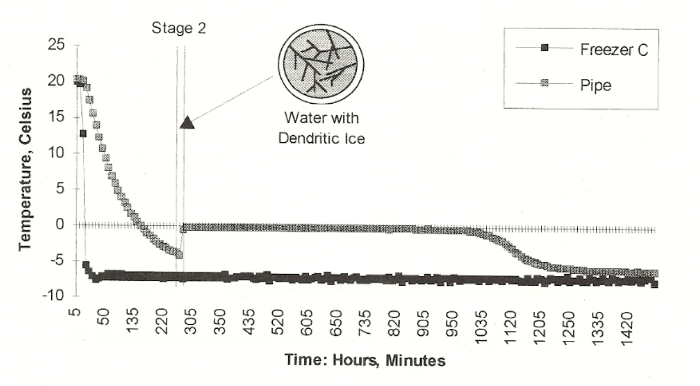

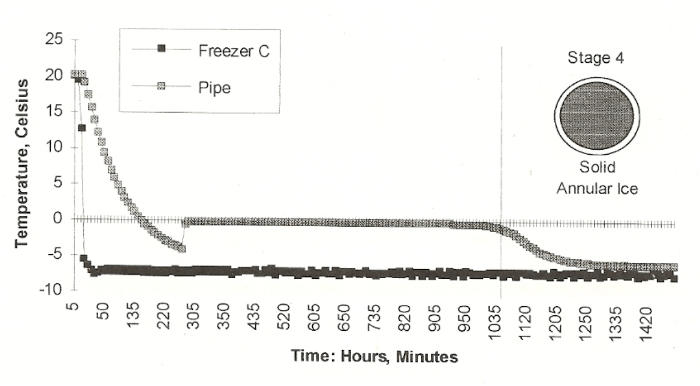
There is captured water downstream from the ice blockage, and the blockage is growing and acting like a piston, compressing the downstream water. If there is no relief, the water pressure will build up until the fluid pressure causes rupture. Jeff took the project from a freezer in the basement to a lab building attic during cold weather. His experience with the freezer taught him that the piston effect is best seen if there is a single spot that is chilled faster than the remainder of the pipe. He devised a fan to blow on the pipe at one spot in the cold attic. The chart below shows what he found.

The results were classic. The attic air temperature supercooled, then flashed to the two-phase condition. The pipe water held exactly 0 degrees for a few hours as annular ice grew inward. At the moment that the temperature began to drop below 0, indicating blockage, the water pressure downstream began to climb. It climbed up to the expected rupture strength of copper, then dropped when the metal left the elastic range, then ruptured, in classic stress-strain behavior.
The lesson is clear: Conventional thinking is that if water pipes are subject to freezing, the water will somehow expand into a larger cylinder of ice and rupture the pipe. Wrong. The rupture is due solely to elevated fluid pressure. Some wonder if ice crystals might form in a way that pushes the pipe walls apart. If this were the case then the stresses would be seen in the ice mass itself. Have you ever seen an ice cube explode? Ice cubes, and the people who chill their drinks with them, are quite relaxed.
A pipe will burst at the weakest location downstream from the blockage. Water supply pipes are designed for all fittings and fixtures to fail at higher pressure than the pipe wall, so the pipe fails. We often see failure at the margin between the ice and water. Ductility of copper is greater at warmer temperatures so we can consider that the pipe fails at its least ductile location. More likely, we can imagine slight temperature fluctuations in the pipe system; at the ice/water margin there might be a succession of events alternating between liquid pressure expansion and ice formation that ratchets the margin area outward. Once the circumference is even slightly enlarged, its strength is reduced at that location, so it becomes a favored leak location.
Are you interested in embarrassing your plumber? Easy. Ask her (or him) if it’s usually the hot water pipe or the cold that needs to be repaired in residential work. Answer: hot water. Then ask why. You’ll get a cockamamie response, like “hot water freezes faster than cold water”. [1] Actually it’s because of the toilet, which acts like a pressure relief device. When the pressure builds up the higher pressure will force the float slightly down in the tank water, allowing a few drops of water out of the pipe and into the tank. It takes only a few drops. The toilet tank protects the entire cold water group. The hot water group doesn’t have this protection.
How to protect against burst plumbing pipes? Any form of pressure relief will do. Leaving the faucet slightly open provides pressure relief. Both hot and cold water sides need to drip. I suppose people think that it is the heat being carried by the leaking water that prevents ice buildup. Wrong. It’s pure pressure relief. Air in the system will work as well. A water hammer arrester can be sized to accommodate a growing blockage of ice.

How to do this in practice? That was the question we tried to answer for State Farm Insurance. Jeff Gordon and I devised and patented a new faucet washer, one that had hard rubber inserts in a softer more compressible ring. The faucet would feel closed against the hard inserts but the soft rubber would compress with elevated water pressure. This was written up in Popular Mechanics magazine, in a piece which opened with “Leave it to researchers at the University of Illinois to invent the leaky faucet.” No buyers, plus, who uses faucet washers these days?
We suggested that State Farm Insurance send a few pressure relief valves to each of their customers, with directions how to install. It is relatively simple to find the hot and cold valve stems that connect the shutoff at the wall to the faucet fixture. Then a compression T is installed in the stem together with the relief valve. The relief valve should be threaded into an appropriate compression T; a non-threaded valve could shoot across the room like a bullet. If the relief valve is doing its job and relieving pressure following blockage, a teaspoon or so of water will drip from the valve onto the floor or the lavatory cabinet. Once again, no buyers.
It would be wonderful to provide burst protection for fire suppression piping. Except that normal measures to prevent freezing of wet fire suppression piping might begin to be relaxed by less-than-conscientious installers. Piping that reaches blockage and is protected—normally by a water hammer arrester rather than a relief valve—will not burst. But it won’t suppress fire either. No buyers. Still, installing a water hammer arrester at a head that is located in a vestibule might not be a bad idea—vestibules are at the greatest risk of pipe freezing.

So. How about we get through a cold spell with no burst water pipes. Can you buy that?
[1] I decided to test this hypothesis that hot water freezes faster than cold for myself. At the time we had an old Hotpoint refrigerator which had an aluminum freezing cabinet up in the corner. I prepared thermocouples to track the water temperature connected to a datalogger running at 1 second intervals. I carefully put the same quantity of hot and cold tap water in each of two ice cube trays, which I carefully placed in the freezing compartment, placed the thermocouples in the water and closed the compartment door and refrigerator door.
And there right before my very eyes, the hot water tray started cooling off quickly and the cold water tray was cooling off more slowly. After a few minutes, the temperatures in the two trays were the same, and then the hot water tray continued its much steeper fall into cold conditions. I had to shake my head. So I saved my data (long lost now), and removed the thermocouples. I removed the cold water tray from the compartment and emptied it. I reached for the hot water tray and it was stuck to the floor of the compartment. Duh.
The aluminum compartment had a layer of frost. The cold tray sat on the frost, and the frost insulated the tray from the refrigerant coursing through the aluminum surround. The hot tray melted it, in effect welding the tray to the compartment.
There are still those who claim that hot water seems to chill faster than cold water. I have strong suspicions that their boundary conditions have little anomalies, just like mine did.

Leave a comment